- The document is supplied as a guideline only, not as a guarantee in itself.
- The aim of this protocol is to find an optimum balance between product safety and commercial effort in order to achieve commercial sterility.
- A common aseptic validation protocol defines what can be expected from an aseptic line.
- All the basic definitions this document refers to are explained in the appendix.
Aseptic Validation Protocol
Preface
Conditions
Plant Pre-requisites
- All relevant equipment operating manuals, recommended maintenance schedules, as well as generally accepted good manufacturing practices etc. are followed.
- The aseptic block needs to be able to run at nominal speed.
- The upstream units need to be able to provide the required quantity and quality of packaging material needed for the microbiological validation.
- The downstream units need to be able to handle the required quantity of filled and closed bottles produced during the microbiological validation in a way that ensures the integrity of the sealed bottles.
- To run the validation, the packaging material must meet the correct specifications. (see appendix 2).
Low Acid versus High Acid Products
- This guideline distinguishes between procedures for low acid products and high acid products (according to the FDA definition).
- Low acid products : pH > 4.6
- High acid products : pH ≤ 4.6
- If a low acid validation is passed, the line is also validated for high acid products, if all parameters of the aseptic block remain the same.
Thermal Treatment of the Product
- As heat treatment for high acid products is not sufficient for killing all kinds of microorganisms including heat resistant spores, the temperature/time regime affects the final results of the validation. It is recommended to apply the highest temperature and longest holding time which the process unit allows for in order to minimize this influence. For explanation of heat treatment see appendix 3.
- It is recommended to detect thermophilic spore formers in the raw product just before the heat treatment as well as in the aseptic storage tank and in the filled bottles.
Sample Size
The validation is based on three runs of the dedicated numbers of bottles and product:
Validation Sequence
Three consecutive days are chosen to run the tests according to the following sequence:
Fill Level of the Bottles
- Half fill the bottles in order to shorten the incubation time because of a higher oxygen level in the headspace.
- If bottle handling (transport, stacking) is critical, the fill level may be increased up to the necessary level e.g. 80% of the nominal volume.
- If nitrogen is used in commercial production, the validation should also include nitrogen dosing.
- If the customer wants to have sellable product, the bottles may be filled with product up to the nominal volume.
Validation Format
- Only one format is chosen for microbiological validation: Usually this is the one with the lowest treatment time available i.e., the smallest bottle because it runs at highest speed and represents the most critical condition.
- In agreement with both parties, also the main contractual format or the one with the most difficult shape or geometry may be selected as the validation format.
- If the microbiological performance has been proven with this format all other formats perform equal or better. In this case, no further microbiological tests with other formats are required.
Validation Medium
- If the intention is to sell the tested production volume once the aseptic system has been validated, the intrinsic quality of the product must follow the customer's specifications.
- For the microbiological validation of low acid aseptic lines a culture medium is used to enable visual inspection. Alternatively, the customer’s product may be used, e.g. UHT milk or others.
- For the microbiological validation of high acid aseptic lines a clear product (e.g. apple juice) is used to enable visual inspection.
Incubation / Evaluation
Storage Conditions
For low acid products
- In case of culture medium or clear product in clear bottles, all 30,000 bottles are visually inspected after 7 days of incubation at a temperature of 30°-32°C.
- In case of turbid product in clear bottles, all 30,000 bottles are visually inspected after 7 days of incubation at a temperature of 30°-32°C. Additionally 1,000 bottles per run are checked for pH.
- In case of non-transparent bottles, 30,000 bottles are visually inspected after 7 days of incubation at a temperature of 30°-32°C. Defective bottles change their size or their shape (gas production or vacuum generation). Additionally 3,000 bottles per run are checked for pH level.
For high acid products
- Half filled or completely filled bottles are stacked on pallets and stored in a secured place at constant temperature of 25 to 30°C for 14 days. All 90,000 bottles are visually inspected.
- Half filled bottles with liquid nitrogen treatment are stored at constant temperature of 25 to 30°C for 14 days. All 90,000 bottles are visually inspected.
- Completely filled bottles with liquid nitrogen treatment are stored at constant temperature of 25 to 30°C for 21 days. All 90,000 bottles are visually inspected.
Evaluation
All packages failing microbiological testing are to be saved for package integrity testing. If any contaminated bottle is found, microbiological identification is carried out in order to determine the origin of the contamination. Especially heat resistant spore identification must be carried out in order to exclude microorganisms coming from the raw material that may have survived the thermal processing.
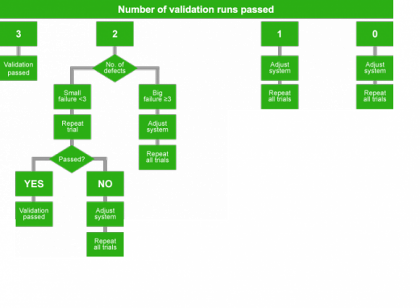
Acceptance Criterion
- The test is accepted if there is not more than one defect bottle per run identified.
- Statistically, zero is not defined. This is the reason why zero defects out of a certain amount of bottles can not be proven.
Acceptance Steps
Appendix
Definitions Aseptic Filling
We agree on aseptic filling in contrast to hot filling and call it

- This technology is the technical precondition which allows aseptic filling of microbiologically sensitive beverages.
- In the process, a pasteurised, sterilised or aseptically filtered product is filled contamination-free into sterilised containers and sealed.
- Finished packs sealed according to this procedure have permanent microbiological stability.
Initial Bioburden of Packaging Material
Microbiologic raw packaging material evaluation:
- Before using packaging materials such as preforms, caps or bottles, the initial bioburden will be evaluated. This evaluation will be carried out on 10 units of the packaging material sampled in accordance with good laboratory practice.
- Preforms or caps will be placed in sterile vials filled with a sterile recovery liquid based on pure sterile water containing 1 ‰ of Tween 80.
- Inside of the bottles: fill 100 ml of the recovery agent in the bottle, and close it with a sterile cap. Complete bottles: cut the bottles in different parts under aseptic conditions in order to be able to place the pieces in a sterile container. Then proceed as with caps or preforms.
- After shaking, the recovery liquid is filtered on a membrane that will be incubated on a Plate Count Agar during 5 days at 30°C.
- After incubation the CFU’s will be counted.
Definition CIP/COP and SIP/SOP
CIP = cleaning in place
CIP means automatic cleaning of internal parts of pipes, vessels, etc. by liquid products (e.g. appropriate chemicals)
COP for us means: cleaning of external surfaces inside an isolator by liquid products (appropriate chemicals)
SIP = sterilization in place
SIP means automatic sterilization of internal parts of pipes, vessels etc. by
appropriate methods
SOP = automatic sterilization of external surfaces inside an isolator by means of
sterilants, disinfectants or other appropriate methods
Integrity Test
Procedure using electrical conductivity
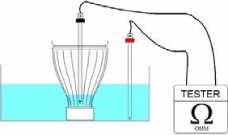
The packaging tightness test is performed to detect potential leaks on bottles closed with caps or aluminium foils using the measurement of the electrical resistance.
Equipment, instruments and reagents used
- City water that has a measurable conductivity
- Tester to measure the resistance. The apparatus needs to reach 2000 MΩ
Prerequisites
- Define the quantity of bottles required for performing the test, at least 100
- Define cap application torque using a specific dynamometer
- Each bottle must be cut without damaging the closure
Procedure
Each bottle must be cut and soaked in standard city water
Introduce inside every bottle approximately 100 ml of the same standard city water
With a tester, set the resistance measurement modality on 2000 MΩ and insert one of the electrodes inside the bottle in contact with the city water and the other one inside the bath outside of the bottle.
Since the two environments, the one inside and the one outside the bottle are separated, the tester shows an infinite resistance called "not readable/out of range".
The first measurement must be carried out 10 minutes after start. The bottles stay in the bath for 72 hours During that period some measurements are performed before the bottles are removed.
If the tester shows a value that is readable there is a connection between the inside and outside of the bottle. This is considered as an untight package.
No leak is accepted in this trial of 100 bottles
Inoculation methods
Preparation of spore suspension
Inoculation methods for packaging material need to be carried out in a professional way in order to achieve results that are indisputable.
General consideration regarding spores
- Spores of bacteria's or moulds should be from a germ which is listed in an official type culture collection and commercially available.
- For spore suspensions, there should be different, independent suppliers available.
- The spores need to be prepared in a water suspension free from salts and organic material that could provide a residue during drying.
- Spore dilutions need to be prepared in pure sterile water. No ringer solution may be used.
- Regarding spores of moulds, they need to be free from organic material coming from cultivation. Normally the spore suspensions are washed 3-4 times and centrifuged in order to remove any kind of foreign matter.
There are three different methods for the inoculation of the packaging material:
- Inoculation with a single dot: The dot has a volume of 10µl and is placed with a micro pipette on the surface of the material that needs to be tested. When starting from a defined concentrated spore suspension (e.g. 108 spores/ml), dilutions will be prepared in order to have the final correct concentration level of spores. The inoculation area will be marked with a circle.
- Inoculation with multi dots: The dots will have a total volume of 10ìl and are placed with a micro pipette on the surface of the material that needs to be tested. When starting from a defined concentrated spore suspension (e.g. 108 spores/ml), dilutions will be prepared in order to have the final correct concentration level of spores. The sum of the individual dots concentration will be equal to the concentration level that is required. If the final level is 10,000 spores and 10 dots are placed on the material, each dot will have an amount of 1,000 spores. The inoculation area will be marked with circles.
- Spray inoculation: A sprayer that delivers a constant volume and a homogeneous fine film will be used to inoculate the inside of the packaging material. The difficulty is to carry out constantly the same distribution of inoculation. Training of the operators needs to be carried out.






.png)